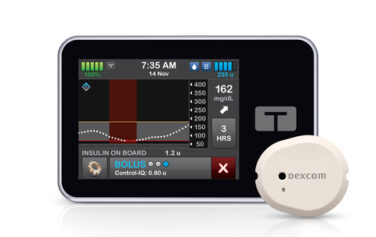
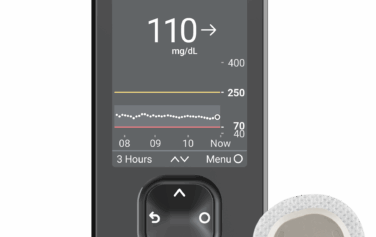
Insulet Omnipod 5
The Omnipod 5 Automated Insulin Delivery System picks up where Insulet’s highly successful Omnipod DASH Insulin Management System left off. Both systems rely on a tubeless and waterproof* Pod to continuously deliver insulin for up to three days. However, only the Omnipod 5 system is automated and designed for use along with leading CGM models like the Dexcom G7 and Freestyle Libre 2+. The Omnipod 5 allows you to take control and avoid highs and lows more effectively than ever.†
The system includes three basic components: the Pod, a compatible smartphone‡ (or a specialized controller, offered free of charge), and the Dexcom G7 system (sold separately).
The Omnipod 5 may be covered by your insurance or Medicare plan. To see if you qualify, start your order or call us at 877-840-8218.
Features
- SmartAdjust™ technology adjusts your insulin delivery rate every five minutes
- Activity mode temporarily reduces insulin levels during workouts
- SmartBolus bolus calculator takes your CGM data and trends into account
- Tubeless, waterproof* Pods - just like those used in the Omnipod DASH system
- Avoid the need for multiple insulin injections each day
- Controllable with a smartphone app‡ or the Omnipod 5 Controller
Download Omnipod 5 User Guide
For all the info you need on the Omnipod 5 Automated Insulin Delivery System, download its user guide. You can also find important safety information on Insulet’s Omnipod 5 website.
FREQUENTLY ASKED QUESTIONS

Medical Review by Shirley DeLeon, Certified Diabetes Care and Education Specialist
*The Pod has an IP28 rating for up to 25 feet for 60 minutes. The controller is not waterproof.
†Brown S. et al. Diabetes Care. 2021;44:1630-1640. Prospective pivotal trial in 240 participants with T1D aged 6 - 70 yrs. Study included a 14-day standard therapy (ST) phase followed by a 3-month Omnipod 5 hybrid closed-loop (HCL) phase. Mean time in range (70-180mg/dL) in adults/adolescents and children as measured by CGM: ST = 64.7%, 3-mo Omnipod 5 = 73.9%, P<0.0001; ST = 52.5%, 3-mo Omnipod 5 = 68.0%, P<0.0001, respectively. Mean time > 180 mg/dL (12AM-6AM) as in adults/adolescents and children, ST vs. 3-mo Omnipod 5: 32.1% vs. 20.7%; 42.2% vs. 20.7%, P<0.0001, respectively. Mean time >180 mg/dL (6AM -12AM) in adults/adolescents and children, ST vs. 3-mo Omnipod 5: 32.6% vs. 26.1%; 46.4% vs. 33.4%, P<0.0001, respectively. Median time in <70 mg/dL (12AM-6AM) in adults/adolescents and children, ST vs. 3-mo Omnipod 5: 2.07% vs. 0.82%,P<0.0001; 0.78% vs. 0.78%, P=0.0456, respectively. Median time <70 mg/dL (6AM-12AM) in adults/adolescents and children, ST vs. 3-mo Omnipod 5: 1.91% vs. 1.08%, P<0.0001; 1.17% vs. 1.62%, P=0.2545, respectively. Results measured by CGM.
‡For a list of compatible smartphone devices visit omnipod.com/compatibility.
Related Articles
Diabetes, Products, Technology
Different Types of CGM: A Complete Guide
Blog, Diabetes, Products, Technology
What are Continuous Glucose Monitoring Devices and How to Choose a CGM?
Blog, Diabetes, Diet & Fitness
Just Diagnosed With Diabetes: A Guide
Blog, Diabetes
Is a CGM FSA Eligible?
Blog, Diabetes, Diet & Fitness
Diabetes in Men: Taking Control of Stress
Blog, Diabetes
Top Medicare Deductible Questions
Blog, Diabetes, Information, Research
Insulin Overdose: Signs, Treatment, and Prevention
Blog, Diabetes, Nutrition
What is Postprandial Blood Sugar?
Blog, Information, Products, Technology
Dexcom G6 vs G7: What to Know
Blog, Diabetes
Does Medicare Cover Diabetes Supplies?
Blog, Diabetes, Information, Research
Can You Trust Your Diabetic Supply Company? 3 Signs to Look For
Diabetes, Diet & Fitness
5 Steps for Managing Diabetes | Type 1 and Type 2
Blog, Diabetes, Information
Prediabetes: Key Risk Factors and Effective Lifestyle Changes
Diabetes, Products, Technology
Comparing the Best Insulin Pumps: Features, Pros, and Cons for Managing Diabetes
Blog, Diabetes, Technology
Insulin Pumps and CGMs: A Complete Guide for Diabetes Management
Blog, Diabetes, Information, Products, Technology
How to Apply a CGM Sensor
Blog, Diabetes
Type 1 Diabetes vs Type 2 Diabetes
Diabetes, Products, Technology
Do I Need a CGM Prescription? Answers for People with Diabetes
Blog, Diabetes, Information, Research
Managing Diabetes and Depression: Strategies for Your Mental Health
Diabetes, Information
Managing Diabetes When You’re Sick: Essential Tips
Diabetes, Information, Research
Can Stress Raise Blood Sugar? | A Guide to Managing Stress and Diabetes
Diabetes, Diet & Fitness, Information, Research
The Link Between Diabetes and Cardiovascular Disease: What You Need to Know
Blog, Diabetes, Information
How to Care for Diabetes: A Guide for Families and Caregivers
Diabetes, Information, Products, Research, Technology
The Future of Diabetes Management: Why CGM and GLP-1s are a Symbiotic Pair
Diabetes, Information, Products, Technology
Best Blood Glucose Meters for 2024 | A Curated Guide
Diabetes, Products, Technology
Dexcom G7 Compatibility with Tandem t:slim X2: What You Need to Know | US MED
Blog, Diabetes, Nutrition
Diabetes and Dental Health: How to Protect Your Smile and Prevent Complications
Diabetes, Products, Research
The Role of Technology in Diabetes Management: From Apps to Continuous Glucose Monitors
Blog, Diabetes
Diabetes and Family Support: Navigating the Impact and Outcome Together
Blog, Diabetes, Diet & Fitness, Nutrition
Intermittent Fasting and Diabetes: Potential Benefits and Consideration
Blog, Diabetes, Information
Having a Social Life and Navigating Events with Diabetes
Blog, Diabetes, Diet & Fitness
Is There a Link Between Diabetes and Stress?
Blog, Products
Main Differences Between the Freestyle Libre 3 vs 2
Blog, Diabetes, Products
What Is a Closed-Loop System?
Blog, Diabetes, Holidays, Information
Traveling with Diabetes: Tips for a Smooth Journey and Blood Sugar Control
Blog, Diabetes, Information
Going Back to School with Diabetes
Blog, Diabetes, Information
What is Diabetic Neuropathy?
Blog, Diabetes, Information, Research
Shoes, Diabetes, and Foot Health
Blog, Diabetes
10 Natural Ways to Combat Diabetes
Blog, Diabetes, Diet & Fitness, Information, Nutrition, Research
Mushrooms and Diabetes: How Can Mushrooms Help with Blood Sugar Levels?
Blog, Diabetes, Information
Diabetes Support Groups: Why Are They Important?
Blog, Diabetes, Diet & Fitness, Exercise
Staying Active with Diabetes: How Yoga for Diabetes Can Help
Blog, Diabetes, Information, Research
How Does Diabetes Affect Sleep?
Blog, Diabetes, Information
Being a Parent with Diabetes
Blog, Diabetes, Nutrition
Pre-existing Diabetes and Pregnancy
Blog, Diabetes, Information, Products, Research, Technology
Dexcom G7 Release Date, Features & Cost Insights
Blog, Diabetes, Products, Technology
Dexom G7 CGM Super Bowl Ad with Nick Jonas
Blog, Diabetes, Information, Products, Research
Know Your Diabetes ABCs | US MED
Blog, Diabetes, Information, Research
Does Diabetes Cause High Blood Pressure?: Understanding Between High Blood Pressure and Diabetes
Blog, Diabetes, Information
Diabetes Sweating Connection: Why Does Diabetes Make You Sweat?

Currently a
US Med® Member?
If you're an existing member, the fastest and easiest way to reorder is by logging into the members portal, where you can also update your account.

Interested in Becoming a US MED® Member?
It's simple, fast and easy! Just complete a quick form or call 1-877-840-8218 now and let us take care of the rest.

Prefer to reorder by phone?
No problem! Just call 866-723-6958 and our agents will walk you through the reorder process.